Menu
KwikCert provides ISO 13485 INTERNAL AUDIT CHECKLIST Document Template with Live Expert Support. By using this document you can Implement ISO 13485 yourself without any support. We provide 100% success guarantee for ISO 13485 Certification. Download this ISO 13485 Documentation Template for free today. This INTERNAL AUDIT CHECKLIST Document Template is part of the ISO 13485 Documentation Toolkit. The toolkit combines documentation templates and checklists that demonstrate how to implement this standard through a step-by-step process. In addition, you can access help from our experts to keep you on the right path, ensuring a straight-forward journey to ISO 13485 certification.
How to create a checklist for an ISO 13485 internal audit for your QMS Author: Waqas Imam One of the most important checking tools in a Quality Management System (QMS) for medical devices, or any management system, is the internal audit. A step by step guide to complying with ISO 13485 and FDA quality management system. A Step-by-Step Guide to Complying with Medical Device QMS Requirements. I am providing you a free QMS audit checklist that includes both FDA 21 CFR part 820 and ISO 13485.
By using this 13485 INTERNAL AUDIT CHECKLIST Document Template, you have less documentation to complete, yet still comply with all the necessary guidelines and regulations. The Documentation Template decreases your workload, while providing you with all the necessary instructions to complete this document as part of the ISO 13485 certification requirement. If yours is a small company looking to implement the ISO 13485 Medical Device QMS by applying the mandatory documents required by ISO 13485 requirements, as well as documenting the common non-mandatory procedures, then this is the perfect template. Using this template ensures you are able to conform to the leading Quality Management System standard: ISO 13485.
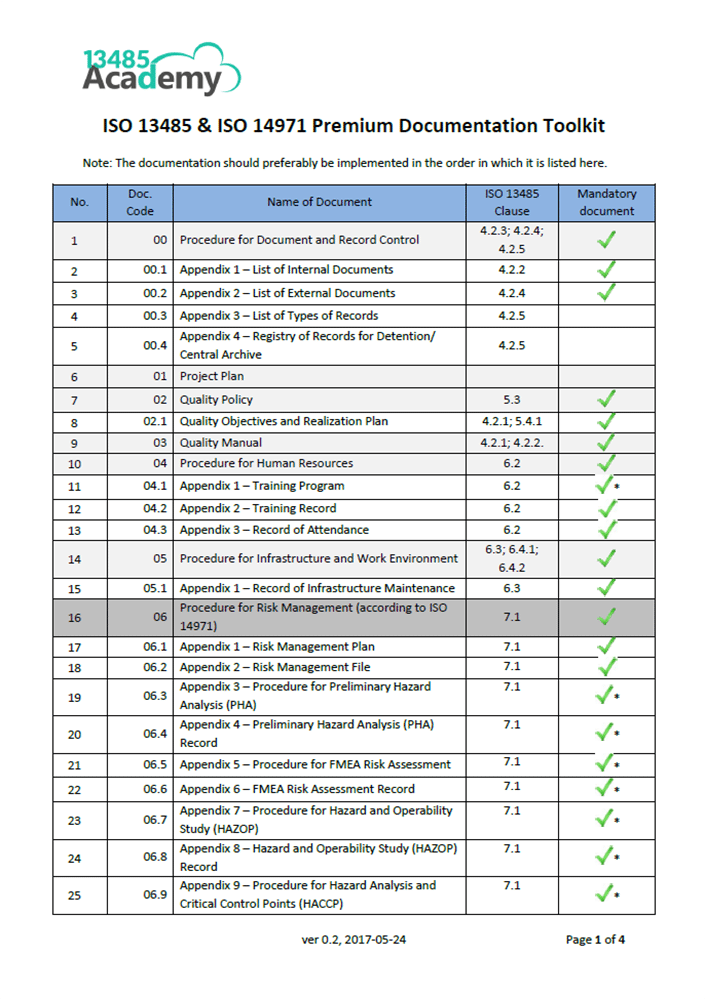
ISO 13485 document template: Internal Audit Checklist The Internal Audit Checklist is the list of questions required to ensure the management system is implemented and maintained. The listing includes more than 100 questions to ensure each requirement of the ISO 13485 standard is implemented and maintained within the Quality Management System, and includes the ability for the company to add additional questions to suit individual needs. The document is optimized for small and medium-sized organizations – we believe that overly complex and lengthy documents are just overkill for you. This document is an appendix to the main document, which is sold separately:. About 13485Academy 13485Academy is one of the Academies of. Advisera specializes in helping organizations implement top international standards and frameworks such as,.
Over the years, Advisera has become a global leader in the provision of web-based training and (information security management) and ISO 22301 (business continuity management). Our products are of best-in-class quality. With a proven performance record of successful implementations in more than 100 countries, our world-class customer support ensures success.
